
How to Move Fast Without Sacrificing Control in Laboratory Systems
The Speed vs. Control Dilemma in Laboratory Systems
Many laboratories face a constant challenge: balancing the need to move quickly – whether to bring new products and assays to market, adopt new technologies, or drive continuous process improvements – while managing the risks associated with the system changes required to support these innovations.
The question is: how can labs evolve their informatics systems quickly without losing control?
In practice, labs often sacrifice speed to protect system validation and maintain regulatory compliance, leading to the view that compliance is the enemy of progress. This perception creates hesitation around making even small system changes, due to concerns about triggering costly and time-consuming revalidation efforts. Over time, this caution becomes ingrained, pushing organizations toward an increasingly conservative approach, delaying updates, slowing the launch of new assays, postponing operational improvements, and ultimately increasing the risk of falling behind more agile competitors.
Why System Changes Feel So Risky
Many legacy LIMS platforms are built on traditional SQL-based architectures that were not designed for frequent change. Updates often require cumbersome schema modifications and database migrations, making even small adjustments complex and time-consuming.
In regulated environments, significant system changes typically require revalidation to demonstrate that the system continues to function as intended. Because validation can be a detailed and resource-intensive process, organizations naturally become cautious about making changes.
The Visibility Problem in Legacy LIMS
Configuration changes in traditional SQL-based systems often lack transparency, making it difficult to clearly understand what changed, when it changed, and which workflows or processes were affected. Because configuration is typically applied at a system level, even small changes can have broad, hard-to-trace impacts across the lab.
Without this visibility, validation teams tend to take the safest possible approach: treating each change as potentially impacting the entire system. The result is widespread revalidation, consuming significant time and resources that could otherwise be directed toward higher-value work.
To manage this uncertainty, many labs bundle changes together and release them as part of large, infrequent validation cycles. While intended to reduce risk, this approach often has the opposite effect: revalidation scope expands, troubleshooting becomes more complex, and unrelated changes are deployed simultaneously.
With limited insight into what actually changed, pinpointing the root cause of issues becomes extremely difficult.
Ironically, efforts to minimize the frequency of change often make updates more complex and riskier.
Modern, workflow-based configuration models offer a different approach. By constraining configuration to the individual workflow level, it becomes much easier to understand the scope and impact of a change, enabling more targeted validation and faster, safer iteration. While system-level configuration can be appropriate in certain scenarios, the lack of granularity and transparency in legacy systems is a key driver of inefficiency and risk in today’s labs.
Lessons from Modern Software Development
Other industries have faced similar challenges when managing complex systems that must evolve over time. In software engineering, the solution has largely been the adoption of structured version control and release management practices.
Modern software development workflows rely on tools and processes that make change visible and manageable. These practices typically include version control systems that track every change, structured releases that group related updates together, and clear change histories that allow teams to see exactly how a system has evolved over time. Developers can also compare different versions of a system to understand precisely what changed between releases.
These capabilities provide important benefits. Teams can quickly understand what was modified, debugging becomes easier because changes are clearly documented, and updates can be delivered in smaller, safer increments rather than large, risky releases.
The key insight is that successful organizations manage change, not avoid change.
Change is managed through structured versioning, allowing systems to evolve while maintaining control and traceability.
From Change Tracking to Configuration Versioning
Most traditional LIMS offer some form of change tracking, but they typically capture individual edits without providing the broader context of a system release.
A new approach is changing how laboratories introduce configuration updates. Instead of treating changes as isolated edits, related configuration updates are grouped together and deployed as a bundled change, which represents a specific configuration version of the system.
This approach provides a clear view of the system’s state at any given moment, along with a structured history of configuration releases. Users can compare versions to understand exactly what changed between updates, making it easier for laboratories to track how their systems evolve over time while maintaining the traceability required in regulated environments.
Configuration versioning introduces several important operational and compliance benefits for laboratories:
- Clear system state
Configuration versioning makes it possible to identify which configuration version was active at any point in time and when it was deployed. The most capable systems include the configuration version active every time data is saved or updated. This clarity makes it far easier to understand system behavior during audits, investigations, or troubleshooting. - Easier change control
Updates can be documented as structured configuration releases, often accompanied by release notes that describe what changed and why. This aligns naturally with the documentation expectations in laboratory change control procedures. - Faster iteration with lower risk
Teams can introduce smaller, incremental updates rather than bundling many changes into large releases. This reduces the risk associated with each update and allows laboratories to improve their systems more continuously. - Better support for validation and audits
Configuration versions provide clear traceability across system updates, making it easier to demonstrate what changed and when. This visibility helps validation teams focus their efforts more precisely and explain system evolution during audits.
Together, these capabilities allow laboratories to move toward continuous system improvement without sacrificing governance or control.
How Labbit Approaches Configuration Change History
Modern laboratory systems need more than simple change tracking, they need a clear, structured way to understand how the system evolves over time. To support this, Labbit has recently introduced configuration change histories, designed to give teams greater visibility and control over configuration updates while simplifying governance and validation.
In Labbit, configuration updates are deployed as changesets. Each changeset represents a configuration version of the system, grouping related updates together into a structured release. This model makes it possible to clearly understand the system’s configuration state at any point in time and provides a complete history of how the system has evolved.
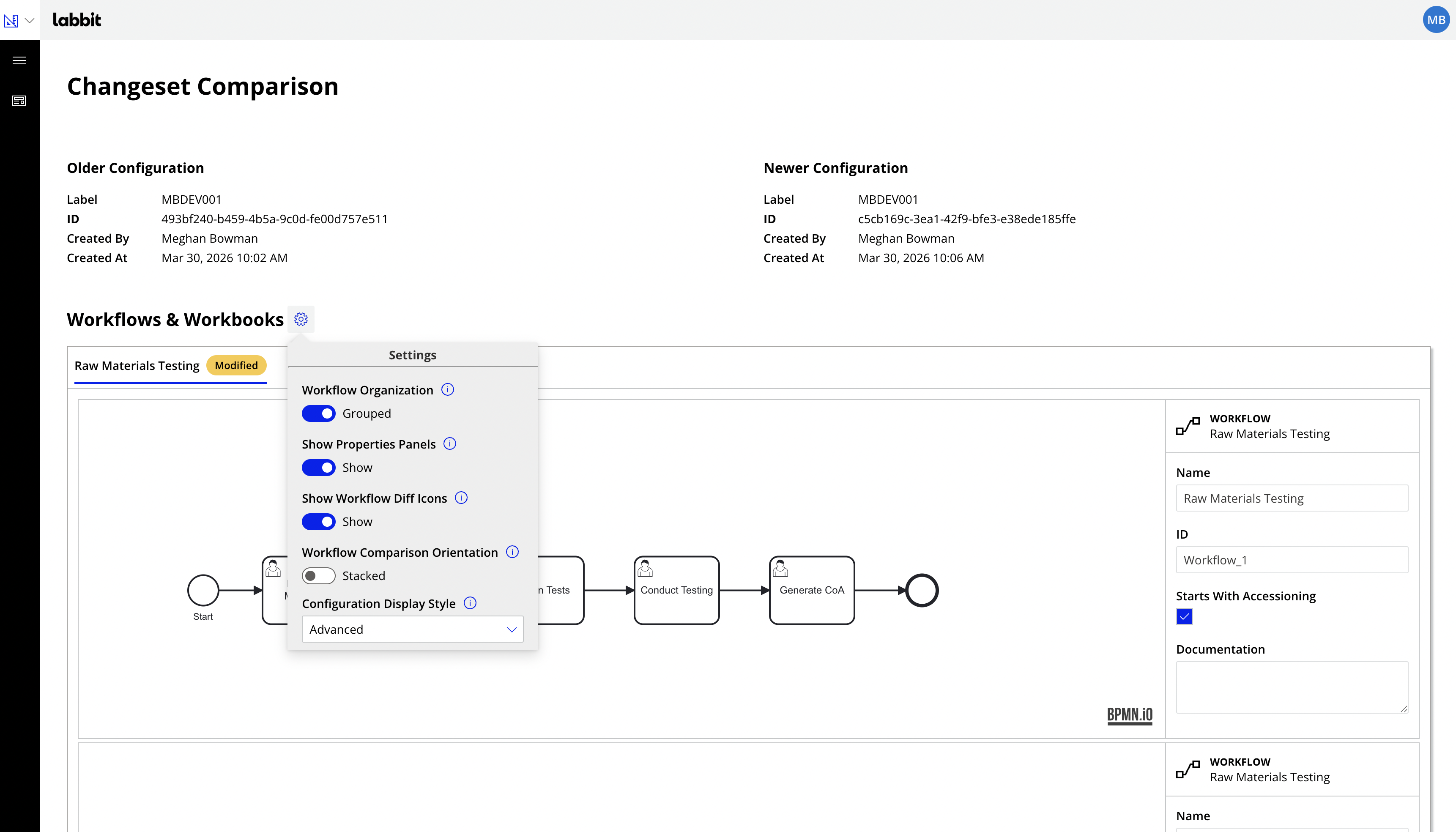
Because each configuration version is tracked and preserved, Labbit provides full visibility into system changes, allowing teams to identify which configuration version was active at any given moment and clearly see the precise differences between any two versions. In addition, every time a workflow is executed, the precise version of the configuration changeset is stamped and viewable on the data, allowing users to clearly see what the system configuration was at any point in time.
This structured approach allows teams to move beyond simple edit tracking and instead view configuration updates as clearly defined releases, much like modern software systems.
Labbit’s configuration change history capabilities are designed to deliver several key benefits.
Gain full visibility and control over configuration changes
Labbit manages configuration through versioned, auditable changesets. Each changeset represents a controlled set of updates that can be reviewed, deployed, and traced with precision, creating an immutable, audit-ready history of how the system evolves over time.
Teams can assess the impact of each changeset before deployment, compare versions to pinpoint issues, and focus revalidation efforts only where necessary, streamlining compliance and enabling more controlled, predictable change management.
Ensure audit-ready configuration traceability
Using Labbit’s graph-based architecture, every configuration changeset deployment is automatically recorded with full context: what was modified, by whom, and when. These immutable, versioned histories eliminate the need to manually reconstruct system changes and provide auditors with clear evidence of controlled configuration management.
Deploy configuration changes with confidence and control
Work with configuration changes as versioned changesets, with continuously updated summaries that make it easy to understand what’s changing and where the impact will be. Before deployment, teams can review and confirm each changeset to prevent unintended updates or disruptions to validated workflows.
If needed, a previous configuration version can be restored seamlessly, enabling controlled, predictable change management without unnecessary risk.
Resolve issues faster and minimize revalidation efforts
Labbit also allows teams to visually compare configuration versions to quickly identify what has changed and isolate the source of issues. This visibility helps quality and validation teams focus their efforts where they are truly needed, reducing investigation time and avoiding unnecessary full-system revalidation.
Together, these capabilities allow laboratories to maintain the governance and traceability required in regulated environments while still evolving their systems more rapidly, helping labs move faster without sacrificing control.
Explore Labbit's environment versioning and configuration histories in this interactive demo.
Conclusion: Continuous Improvement Without Losing Control
Laboratory systems must evolve as science evolves. New assays are introduced, workflows change, and technologies advance. Digital systems need to keep pace with these changes if laboratories are to remain competitive and responsive to new opportunities.
But evolution does not have to mean increased risk.
By treating configuration updates as structured versions, laboratories gain the ability to introduce improvements in a more controlled and transparent way. Teams can implement updates more frequently, maintain clear traceability of system changes, support validation and audit requirements, and ensure that their systems remain aligned with real laboratory workflows as they evolve.
In this model, change is no longer something to avoid, it becomes something that can be managed with confidence.
When laboratories are able to evolve their systems without the fear of disruptive validation cycles or opaque configuration changes, innovation and modernization are no longer hindered. Instead, organizations can introduce new assays faster, adopt emerging technologies more readily, and continuously improve operations—allowing them to stay ahead of the market while maintaining the governance required in regulated environments.
Labbit’s introduction of configuration change histories is an important step in that direction. By bringing greater visibility, structure, and control to configuration updates, laboratories can begin to rethink how they manage system evolution and validation.

And this is only the beginning. As Labbit continues to build on these foundations, the way laboratories approach configuration change management – and the validation processes that support it – is poised for significant transformation. Stay tuned!
Curious to learn more? Watch our webinar, Configuration Versioning for LIMS: Bringing Software-Style Releases to Laboratory Systems.





