
Cell & gene Therapy LIMS
Digitally Transform Cell & Gene Therapy Operations
Trusted By
Key Benefits of Labbit
Build a connected, workflow-driven foundation for scalable, compliant cell and gene therapy laboratory operations.
From Paper to Controlled Digital Workflows
Replace manual, error-prone processes with fully digital, enforced workflows across lab and manufacturing.
Unified Data Across Lab, Manufacturing & QC
Break down silos with a single platform that connects process data, QC results, and manufacturing execution in one structured system.
Real-Time Insights for Time-Critical Decisions
Turn operational data into immediate, actionable insight, especially for release decisions tied to viability and stability constraints.
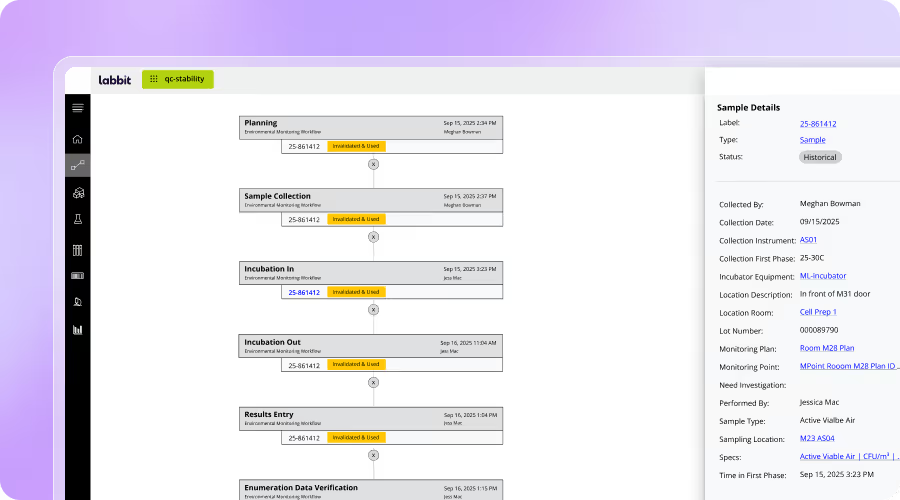
End-to-End Traceability & Chain of Custody
Track every sample, batch, and transformation step with full lineage, custody, and environmental history.
Compliance and Governance by Design
Ensure audit trails, electronic signatures, and data integrity are enforced natively across all workflows.
Scalable Foundation for CGT Growth
Support increasing manufacturing complexity, patient volumes, and regulatory maturity without re-architecting systems.
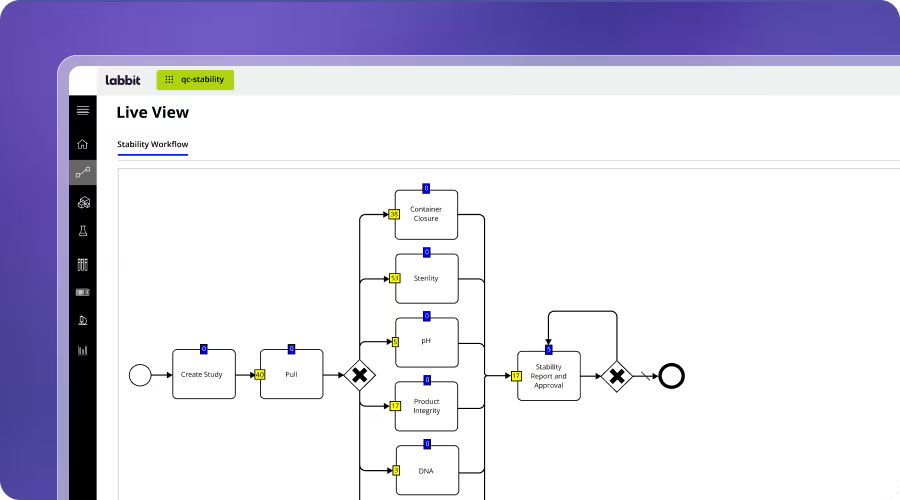
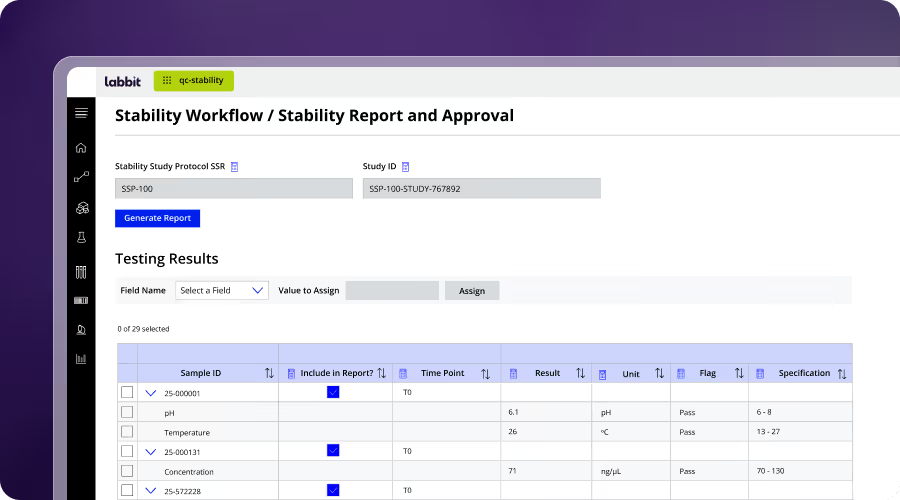
QC & Release Testing
Labbit replaces fragmented QC with workflow-driven execution that ensures consistency, traceability, and compliance. Testing is guided, data is captured in context, and specifications are applied automatically. QC is also tied to stability windows, ensuring results reflect release eligibility.
GMP Manufacturing
Labbit unifies GMP manufacturing through workflow-driven execution of batch records, in-process controls, and deviations. Every step is traceable and time-bound, keeping production within validated and stability windows while enabling compliant manufacturing.
Scale-up & Validation
Labbit replaces informal processes with standardized, traceable workflows. End-to-end lineage and automated audit trails improve transparency across execution, accelerating system validation while ensuring scale-up remains consistent, compliant, and reproducible.
Key features for Cell & gene Therapy Labs
Workflow-First Configuration Engine
Model complex CGT processes—including manufacturing, QC, and stability constraints—as controlled, executable workflows.
Unified Lab & Manufacturing Data
Connect lab, manufacturing, QC, and stability data in a single structured system designed for traceability and analytics.
Stability & Viability Management
Embed time-, condition-, and process-based constraints directly into workflows to ensure therapies remain within validated viability windows across manufacturing, QC, and transport.
Electronic Batch Records (EBR)
Digitally execute and document batch processes with full traceability across materials, steps, and environmental conditions.
Deviation, CAPA & Investigation Workflows
Automatically trigger and manage quality events within execution workflows, maintaining compliance without disrupting operations.
Integrated AI and Analytics-Ready Data Capture
Capture structured, contextual data at the point of execution to enable real-time insights and faster decision-making.
CUSTOMER SPOTLIGHT
Helix
“As we look ahead, we’re always asking how quickly we can iterate—how quickly we can release updates to a workflow or create new ones. Labbit is a key part of that, enabling us to test, validate, and bring new workflows into production more efficiently.”
Tyler Cassens
Senior Product Manager, Helix
Regulatory Compliance + Data Security
Labbit is built for the regulatory rigor of cell and gene therapy manufacturing, where patient safety depends on compliance and timing. Workflows run in a controlled, validated system with audit trails, electronic signatures, and role-based access, ensuring GMP and 21 CFR Part 11/211 alignment.
Unlike fragmented point solutions, Labbit unifies LIMS, LES, and EBR into one system. Stability and viability constraints are embedded in workflows, ensuring compliance is enforced during execution, not after the fact.

Getting Started
See how Labbit works for labs like yours
Book a 30-minute demo to see visual workflow modeling, real-time execution visibility, and knowledge graph insights in action. We'll show you how Labbit adapts to your specific workflows — without the vendor lock-in.






