
Pharma R&D LIMS
Unified, workflow-driven R&D for modern pharmaceutical development
Trusted by




















Key Benefits of Labbit
Turn disconnected lab activity into structured, actionable R&D intelligence
End-to-End Scientific Traceability
Connect experiments, samples, and results across discovery and development with full lineage and contextual metadata.
Reduced Manual Data Handling
Eliminate Excel-driven workflows and transcription errors through automated, structured data capture.
Unified Data Across Teams
Break down silos between groups with a shared operational and scientific data model.
Full Pipeline Visibility
Understand what’s happening upstream and downstream to make better-informed decisions in real time.
Improved Reproducibility
Standardize workflows and capture experimental context to ensure results can be consistently replicated.
AI-Ready Scientific Data
Generate structured, machine-readable datasets that support analytics, modeling, and AI applications.
Screening
Early-stage screening teams operate in high-throughput environments where fragmented data slows decisions. Labbit enables structured experiment design, automated assay capture, and consistent metadata tracking across campaigns. This makes screening data immediately usable, comparable across studies, and connected to downstream workflows, reducing rework and improving hit quality.
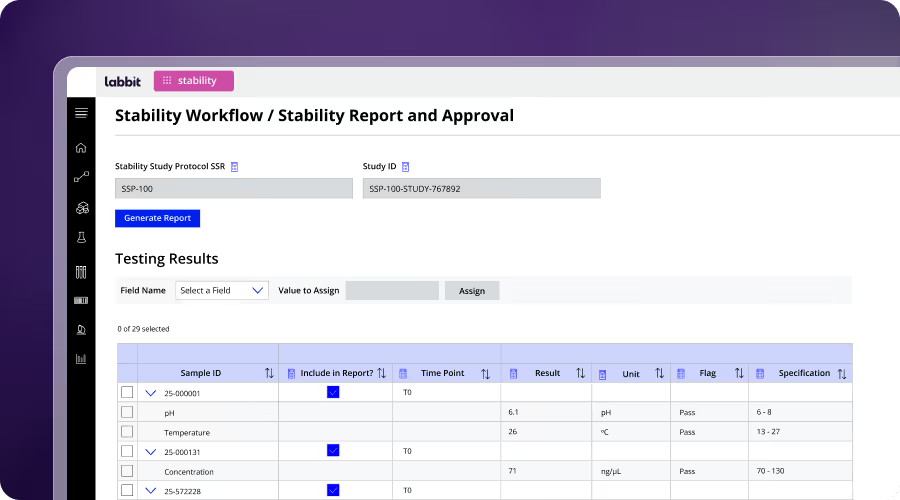
Bioprocessing
Bioprocessing teams manage complex, multi-step workflows across cell lines, bioreactors, and purification processes. Labbit connects process execution, instrument data, and sample lineage in a single workflow system. This enables real-time visibility into process conditions, improves reproducibility, and ensures critical variables like culture conditions and timing are consistently captured and traceable.
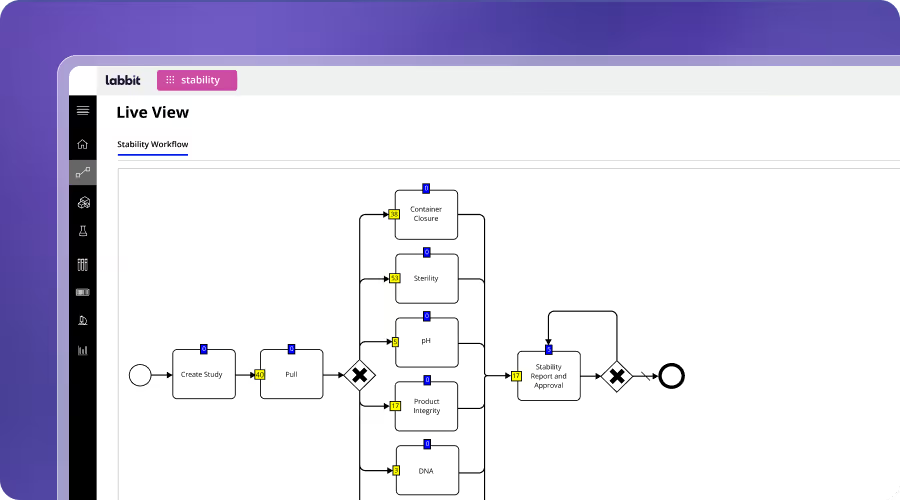
CROs & CDMOs
CROs and CDMOs require standardization, traceability, and flexibility across many concurrent client projects. Labbit enables configurable, workflow-driven execution across multi-tenant environments while maintaining data integrity. It supports seamless handoffs, consistent reporting, and unified data models, improving efficiency and regulatory readiness at scale.
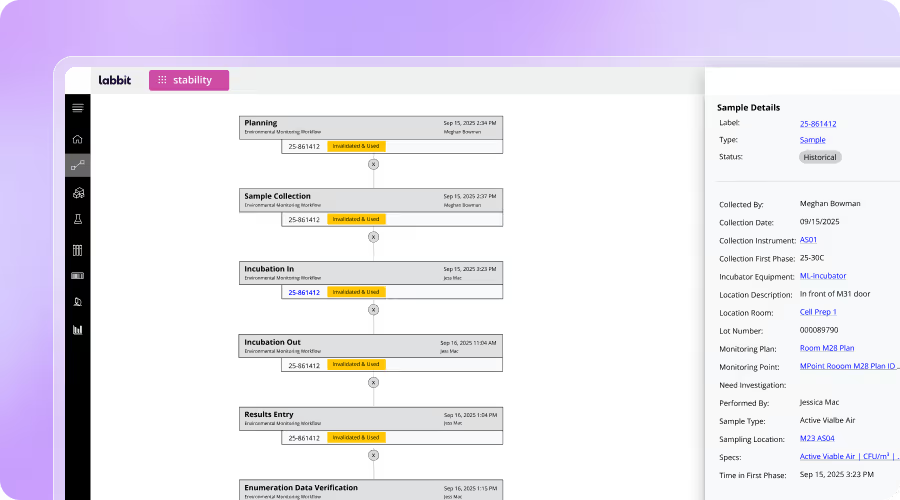
Key Features for Pharma R&D Labs
Workflow-Driven QC Execution
Digitize and enforce lab processes—from sample login to release—ensuring consistency across analysts, shifts, and sites.
End-to-End Sample & Data Traceability
Capture complete lineage across samples, tests, retests, and results with fully contextualized histories.
Automated Data Capture & Integration
Integrate with instruments and upstream systems to eliminate manual entry and ensure data integrity.
Investigation & Deviation Management
Track out-of-spec results, retests, and investigations with full context and audit-ready documentation.
COA & Reporting Automation
Save time and eliminate errors—automate COA and report generation directly from Labbit for precise, compliant results.
Configurable, Future-Proof Platform
Adapt workflows, data models, and processes without code—so your system evolves with your operations.
How Helix Replaced a Legacy LIMS with Labbit to Enable Scalable, High-Performance Laboratory Operations
CUSTOMER SPOTLIGHT
Helix
“As we look ahead, we’re always asking how quickly we can iterate—how quickly we can release updates to a workflow or create new ones. Labbit is a key part of that, enabling us to test, validate, and bring new workflows into production more efficiently.”
Tyler Cassens
Senior Product Manager, Helix
Regulatory Compliance + Data Security
Labbit supports the regulatory rigor of pharmaceutical R&D with built-in audit trails, electronic signatures, and role-based access controls to meet 21 CFR Part 11 and GxP requirements.
By embedding compliance into workflows, every action is time-stamped, traceable, and linked to its process, ensuring data integrity by design. This improves inspection readiness, reduces validation burden, and keeps data secure, consistent, and defensible across the organization.

Getting Started
See how Labbit works for labs like yours
Book a 30-minute demo to see visual workflow modeling, real-time execution visibility, and knowledge graph insights in action. We'll show you how Labbit adapts to your specific workflows — without the vendor lock-in.